Copy link
Cardiovascular Changes with Aging
Last updated: 01/03/2023
Key Points
- Changes in the cardiovascular system with aging include: decreased response to β-receptor stimulation; stiffening of myocardium, arteries, and veins; autonomic nervous system changes (↑sympathetic activity; ↓parasympathetic activity); conduction system changes; defective ischemic preconditioning.
- It is important to differentiate between normal physiological changes with aging and pathologic states such as atherosclerosis. While increased age is a risk factor for many pathologies, not all aging adults develop pathologic changes.
Decreased Response to β-receptor Agonism/Stimulation
- The mechanism of this change is due to a defect in the intracellular coupling within the receptors, not downregulation.1
- This decreased cardiac response to β-receptor agonism is responsible for the decreased efficacy of the cardiac baroreceptor reflex, which results in the inability to increase heart rate in response to hypotension.1,2
- Heart rate and contractility increase less in response to the endogenous release of catecholamines, which accounts for decreased exercise capacity and decreased maximal heart rate with exertion.3
Stiffening of Myocardium, Arteries, and Veins
Decreased ventricular compliance of myocardium
- Cardiac myocytes are terminally differentiated and therefore form no new cells with aging. To compensate for normal cell death, cardiac myocytes undergo hypertrophy. Fibroblasts continue to function with age and produce collagen, which decreases ventricular compliance.4
- Decreased ventricular compliance leads to diastolic dysfunction and ↑LVEDP (left ventricular end-diastolic pressure). As previously mentioned, elderly patients have decreased response to β-receptor agonism. Therefore, ventricles depend more on an adequate end-diastolic volume (EDV) to produce enough contractile strength via the Frank-Starling relationship.3
- With decreased ventricular compliance, older patients rely more on active atrial contraction during late diastole for ventricular filling. Their dependence on this “atrial kick” and the increased diastolic time required for adequate filling is why tachycardia is poorly tolerated in elderly patients.2
- Summary: ↓β-receptor responsiveness → ↓attainable cardiac output (CO) →↑LVEDV to maintain contractility via Frank-Starling length-tension relationship → ↑LVEDP (due to ↓ventricular compliance) → ↑reliance on “atrial kick” and increased diastolic time required for adequate ventricular filling and CO (Figure 1).
Vascular Changes to Arteries and Veins
- Vascular stiffening occurs with age due to several mechanisms, including increased deposition of lipids, decreased elastin production and collagen turnover (leading to intimal hyperplasia), vascular calcification, and endothelial cell dysfunction.2
- These deposits also lead to irregularly shaped endothelial cells, which promote turbulent flow and predispose vessels to further deposition of lipids, or disruption/dissection.2
- Vascular wall stiffening leads to decreased arterial/venous capacitance, which causes pulse waves (which are normally reflected by branch points and arterial walls), to return earlier in the cardiac cycle, resulting in increased ventricular afterload, resulting in worsening left ventricular hypertrophy.1
- Endothelial dysfunction leads to decreased nitric oxide production, resulting in decreased vascular relaxation.2
- All these vascular changes (intimal thickening, larger artery dilation, central artery wall stiffening) predispose to hypertension, increased pulse wave velocity, widened pulse pressure, and put patients at higher rates of cardiovascular disease.2,4
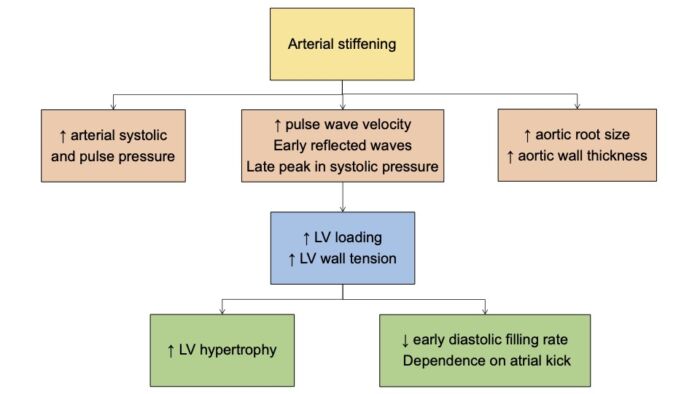
Figure 1. Cardiovascular changes with aging. LV = left ventricular
Autonomic Nervous System Changes
- Patients develop decreased β-receptor function with age and reduced baroreceptor sensitivity which decreases the aging heart’s response to sympathetic stimulation. The mechanism of impaired baroreceptor efficiency is due to a decrease in the heart’s response to stimulation rather than a decrease in the baroreflex control of vascular tone.1
- With aging, there is increased sympathetic activity, while parasympathetic activity remains the same. The increased sympathetic activity results in more circulating levels of norepinephrine (due to increased secretion and decreased metabolism/uptake), which leads to larger swings in blood pressure (mainly due to α-agonism) and a higher incidence of arrhythmias.1,3
Conduction System Changes
- The sinoatrial node and other components of the conduction system undergo a decline in cell numbers, which are replaced with fibrosis and fat. This change in tissue composition is a possible etiology for increased arrhythmias (especially sick sinus syndrome, tachyarrhythmias/bradyarrhythmias) in the elderly.2
- Atrial hypertrophy (possibly developed from the necessary “atrial kick”) is associated with an increased rate of atrial fibrillation.2
Defective Ischemic Preconditioning
- Ischemic preconditioning is the phenomenon in which a brief period of myocardial ischemia will lessen the myocardial damage associated with a subsequent, more prolonged ischemic event. This is commonly mentioned in conjunction with “warm-up angina” in which patients who exercise to the point of angina, rest, and then exercise again can achieve higher levels of exertion before developing another bout of angina. Patients are also less likely to develop heart failure or die of myocardial infarction (MI) if they experience angina within 48hrs of their MI.1,2
- Aging is associated with the loss of ischemic preconditioning, and warm-up angina is nonexistent beyond the age of 75. The preconditioning/cardioprotective effects of volatile anesthetics are also thought to decrease, if not be completely abolished, with advanced age.1,2
References
- Barash PG, Cullen BF, Stoelting RK, et al. Cardiovascular aging. In: Clinical Anesthesia. 8th ed. Philadelphia, PA. Wolters Kluwer; 2017:2241-45.
- Reves JG, Barnett SR, McSwain JR, Rooke GA, eds. Geriatric Anesthesiology. 3rd Ed. Springer; 2017; 1-510
- Alvis BD, Hughes CG. Physiology considerations in geriatric patients. Anesthesiol Clin. 2015;33(3):447-456. PubMed
- Barnett SR. Manual of Geriatric Anesthesia. 1st Ed. Springer; 2013; 1-441.
Other References
Copyright Information

This work is licensed under a Creative Commons Attribution-NonCommercial-NoDerivatives 4.0 International License.