Copy link
Hepatic Blood Supply and Regulation of Hepatic Blood Flow
Last updated: 08/02/2023
Key Points
- The liver has a dual supply from both the portal vein (PV) and the hepatic artery (HA).
- Splanchnic blood flow (SBF) functions to meet the metabolic demands of the gastrointestinal (GI) tract while acting as a reservoir for the circulating blood volume.
- Blood flow through the liver is subject to autoregulation – the hepatic artery buffer response (HABR) which maintains HBF despite any variability in splanchnic blood flow.
Introduction
- Around 25% of the cardiac output goes to the liver, and at any one time, 10% of the cardiac output is held in the liver.
- The liver has a dual blood supply: 75% of the blood is supplied from the PV and 25% from the HA.1,2
- Hepatic sinusoids are the site at which portal and arterial blood inflow interacts with the hepatocytes before draining to the central veins.1
- Both intrinsic and extrinsic mediators of the splanchnic circulation and hepatic blood flow (HBF) are responsible for maintaining normal liver function and normal circulating blood levels.2,3
Hepatic Blood Supply
Inflow: PV1-3
- The PV is formed from the confluence of the splenic vein and the superior mesenteric vein, which usually occurs just posterior to the neck of the pancreas.
- It provides 75% of the blood flow and 50% of the oxygen to the liver.
- The division of the PV into right and left branches occurs outside the liver at the porta hepatis (Figure 1).
Inflow: HA
- The celiac trunk, which arises off the aorta, gives rise to the left gastric artery, the splenic artery, and the common hepatic artery. The common hepatic artery in turn divides into the proper hepatic artery and the gastroduodenal artery.
- The HA provides 25% of the blood flow and 50% of the oxygen to the liver.
- For patients who have normal arterial anatomy, the division of the HA into the right and left branches occurs outside of the liver, at the porta hepatis.
Extrahepatic Portal Triad
- The extrahepatic portal triad, located at the porta hepatis, is where the arterial, portal and nerve supply enter and the common bile duct exits the liver.
- It is located within the hepatoduodenal ligament between the caudate and quadrate lobes.
- Because of its extrahepatic location, it allows surgeons easy access to control bleeding during liver surgery. Cross clamping the hepatoduodenal ligament is referred to as the Pringle maneuver.
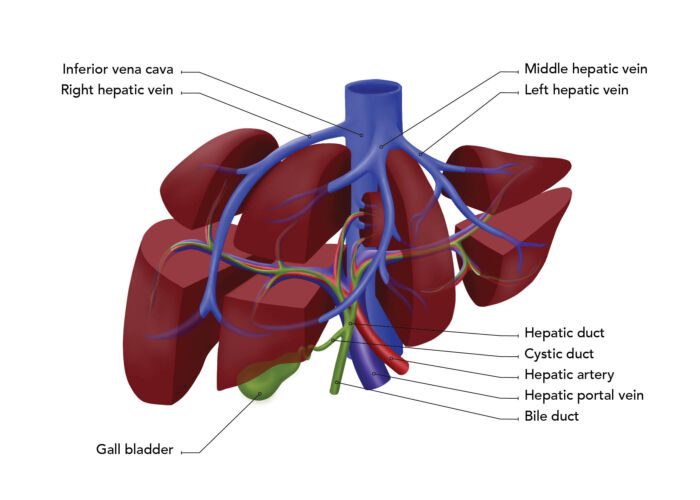
Figure 1. Segmental anatomy of the liver
Intrahepatic Circulation
- Intrahepatic portal triad consists of a branch of the HA, PV, and hepatic duct.
- Multiple triads are located around the periphery of hepatic lobules.
Sinusoids4
- The sinusoids are the site of communication between the arterial and portal inflow, hepatocytes, biliary system, ducts and venous outflow (Figure 2).
- Sinusoids are lined by liver specialized endothelial cells (LSECs). Their fenestration and lack of basement membrane allows for rapid transmission of blood and metabolites into the sinusoids.
- LSECs also have intrinsic vasodilators, the most important of which is nitric oxide. When released, NO stimulates vasodilation of the hepatic sinusoids. Dilated hepatic sinusoids maintain low portal pressure within the liver. Low portal pressure creates a gradient for blood to easily shift from the portal circulation to the systemic circulation.
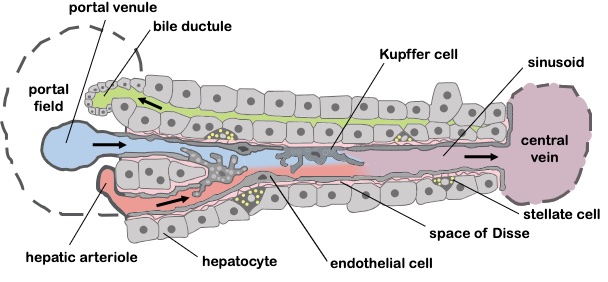
Figure 2. Liver sinusoid. Source: Wikimedia Commons. CC By 2.5. Link. Originally by Frevert U, Engelmann S, Zougbédé S, Stange J, Ng B, et al. Converted to SVG by Viacheslav Vtyurin.
Outflow1
- At the center of each liver lobule is a central vein which receives the mixed blood from the sinusoids.
- Blood entering the central vein flows into intrahepatic veins, which in turn drain into the hepatic veins.
- There are three hepatic veins: the right and middle hepatic veins drain blood from the right lobe while the left hepatic vein drains blood from the left lobe. The hepatic veins drain directly into the suprahepatic inferior vena cava (Figure 1).
Splanchnic Circulation2
- The role of the splanchnic circulation is to provide enough blood flow to meet the metabolic demands of the gastrointestinal tract while acting as a reservoir to hold and release more blood volume to the core circulation when needed (for example, during times of exercise or stress).
- The volume of blood within the splanchnic circulation is dependent on:
- Intrinsic Factors
- A build-up of tissue metabolites (H+, CO2, adenosine, K+) during poor perfusion leads to vasodilation.
- Increased vascular transmural pressure, during an increase in blood flow, leads to reflex smooth muscle contraction and vasoconstriction.
- Extrinsic Factors
- Autonomic nervous stimulation:
- α-1 adrenergic mediated vasoconstriction
- ß-2 adrenergic mediated vasodilation
- parasympathetic mediated smooth muscle relaxation and increases in blood flow
- Autonomic nervous stimulation:
- Humoral Factors
- Locally produced and circulating vasodilatory substances – bradykinin, serotonin, adenosine, peptide hormones, prostaglandins;
- circulating vasoconstrictors – vasopressin, angiotensin II, prostaglandins, neuropeptide Y, etc.; and
- specific food metabolites, such as glucose and long chain fatty acids, also stimulate vasodilation.
- When dilated (usually 30-60 minutes postprandial), the splanchnic circulation can hold 30% of the circulating blood volume within its vessels.
- When stimulated by alpha agonists, vasopressin and angiotensin II, the splanchnic circulation constricts, sending more blood into the portal vein and into the hepatic sinusoids. The extra blood volume will enter the central vein and be deposited into the core circulation via the suprahepatic IVC.
- This transference of extra blood flow from the splanchnic circulation to the core circulation allows the body to increase its core circulating blood volume during times of stress or increased myocardial demand.
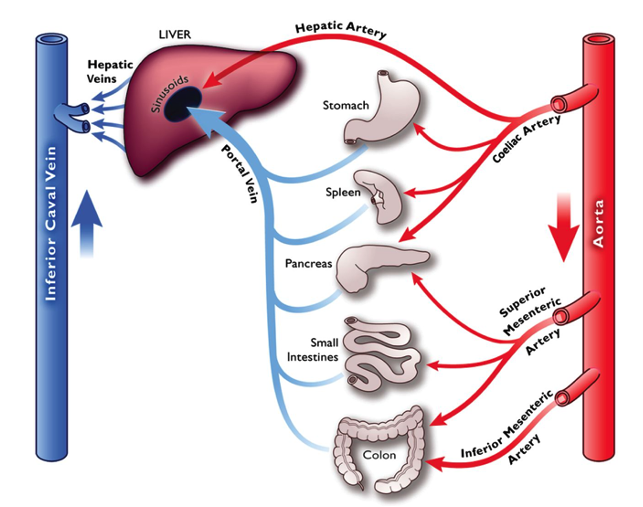
Figure 3. Splanchnic circulation. Used with permission from Harper D, Chandler B. Splanchnic circulation. 2016; BJA Educ.16(2): 66-71. Link.
Hepatic Artery Buffer Response
- A constant, reliable flow through the liver allows efficient function.
- Hepatic blood flow is a function of both HA and PV flow.
- The PV is supplied from the splanchnic circulation, which in turn is dependent on changes in metabolic demands from the gastrointestinal tract.
- The PV cannot regulate its blood flow, therefore all control rests with HA flow.3
- HABR refers to the ability of the hepatic artery to dilate in response to drops in portal flow in order to maintain a constant hepatic blood supply.
- It is mediated by adenosine and can buffer a fall of up to 60% in portal flow.5
References
- Trefts E, Gannon M, Wasserman DH. The liver. Curr Biol. 2017;27(21): R1147-51. PubMed
- Harper D, Chandler B. Splanchnic circulation. 2016; BJA Educ.16(2): 66-71. Link
- Lautt WW. Mechanism and role of intrinsic regulation of hepatic arterial blood flow: hepatic arterial buffer response. Am J Physiol. 1985; 249(5 Pt 1): G549-56. PubMed
- Poisson J, Lemoinne S, Boulanger C, et al. Liver sinusoidal endothelial cells: Physiology and role in liver diseases. J Hepatol. 2017;66(1):212-27. PubMed
- Eipel C, Abshagen K, Vollmar B. Regulation of hepatic blood flow: the hepatic arterial buffer response revisited. World J Gastroenterol. 2010;16(48):6046-57. PubMed
Copyright Information

This work is licensed under a Creative Commons Attribution-NonCommercial-NoDerivatives 4.0 International License.