Copy link
Hyponatermia and Transurethral Resection of Prostate Syndrome
Last updated: 01/15/2024
Key Points
- Hyponatremia is defined as a serum sodium (Na) less than 130 mEq/L. Normal sodium levels range from 135 to 145 mEq/L.
- Hypo-osmolality is defined as a serum osmolality less than 270 mOsm/L. Normal serum osmolality ranges from 285 to 290 mOsm/L.
- The primary driver of serum osmolality is serum sodium, as seen in the following calculation for serum osmolality: (2xNa +Glucose/18 +urea/2.8)
- Transurethral resection of prostate (TURP) syndrome refers to a constellation of symptomatic hyponatremia, excessive intravascular volume, and edema resulting from absorption of hypotonic irrigation fluid during TURP, transurethral resection of bladder, ureteroscopic, or hysteroscopic procedures.
Introduction
- Sodium is the dominant extracellular cation and normally accounts for almost all the osmotically active solutes in the body.1 Normal serum sodium ranges from 135 to 145 mEq/L.
- Only 10% of total body sodium is intracellular, and the concentration gradient between the intracellular and extracellular components is maintained by the Na-K- ATPase pump.1
- Normal body osmolality ranges from 285 to 290 mOsm/kg and is the same in the intracellular and extracellular compartments, secondary to the free movement of water between the compartments.1
- Serum osmolarity = (2 x Na) + (glucose/18) + (urea/2.8)
Etiology
- Hyponatremia is the most common electrolyte disturbance among hospitalized patients.2 Hyponatremia can occur when the serum osmolality is high, normal, or low (Figure 1). Total body sodium must be considered in cases with low serum osmolality.
- An assessment of sodium level, serum osmolality, as well as total body water (or total body sodium) is necessary to accurately diagnose the underlying cause of the hyponatremia. A thorough workup of hyponatremia in the perioperative setting is warranted, as even mild cases have been associated with an increase in 30-day mortality, major cardiac events, wound healing, and pneumonia.1
- In most cases of hypo-osmolar hyponatremia, total body sodium is normal or increased.2
- Postoperative hyponatremia is seen in 1-5% of patients and can occur for multiple reasons, including the surgical stress response, pain and stress-related sympathetic activity, periods of hypotension, etc., resulting in sodium and water retention similar to syndrome of inappropriate antidiuretic hormone (SIADH). Administration of dextrose-containing hypotonic fluids further exacerbates the risk of hyponatremia.1
- In anesthesia practice, hyponatremia classically occurs during transurethral resection of prostate (TURP) or during prolonged ureteroscopic or hysteroscopic procedures.1
- Pseudohyponatremia is defined as a serum sodium less than 135mEq/L in the setting of normal serum osmolality. Hyperglycinemia secondary to the use of glycine-containing irrigation fluids during cystoscopy or TURP procedures is a common intraoperative cause of pseudohyponatremia.3
- Other nonsodium solutes, such as glucose, mannitol, ethanol, methanol, ethylene glycol, and extreme hyperlipidemia and hyperproteinemia can be causes of pseudohyponatremia. These nonsodium solutes can additionally elevate the serum osmolality above normal.
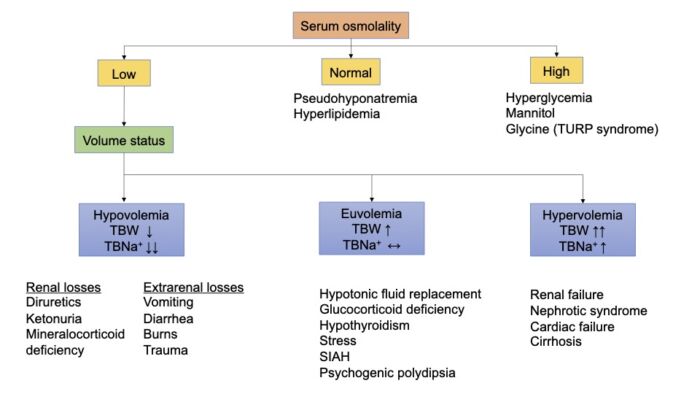
Figure 1. Etiology of hyponatremia. Adapted from Edwards M, Grocott M. Perioperative fluid and electrolyte therapy. In: Miller RD, editor. Miller's Anesthesia. 9th edition. Philadelphia, PA; Elsevier/Saunders; 2019: 1774-5, 1787-90.
Clinical Presentation and Diagnosis of Hyponatremia
- The clinical presentation depends on how rapidly the hyponatremia occurred and the severity of hyponatremia. The symptoms are secondary to cerebral edema and raised intracranial pressure and can range from nausea, headache, and confusion to lethargy, coma, and seizures.1,2
- In the acute setting, symptoms typically occur when the sodium concentrations are less than 120-125 mEq/L (higher in children and premenopausal females). In the chronic setting, patients may be asymptomatic at sodium concentrations less than 120 mEq/L.1
- An assessment of sodium level, serum osmolality, and total body water (or total body sodium) is necessary to accurately diagnose the underlying cause of the hyponatremia in a stepwise fashion (Figure 1). The steps of assessment are:
1. determine the level of hyponatremia;
2. determine the serum osmolality; and
3. assess the patient’s total volume status.
TURP Syndrome
- TURP is a common urologic procedure for benign prostatic hypertrophy in men over age 65. It is performed using a modified cystoscope (resectoscope) with a wire loop connected to an electrocautery unit (monopolar or bipolar).
- TURP may be performed under general or neuraxial anesthesia. Spinal anesthesia may reduce the risk of pulmonary edema and allows the early detection of mental status changes with TURP syndrome (see below).
- Typically, during the procedure, large amounts of irrigation fluids are absorbed into the circulation or more slowly from the retroperitoneal and perivesical spaces. The amount of irrigation fluid absorbed depends on the volume and pressure of the irrigation, duration of resection, intravesical pressure, and the degree of bleeding.
- TURP syndrome refers to a constellation of symptomatic hyponatremia, excessive intravascular volume, and edema resulting from absorption of hypotonic irrigation fluid during TURP, transurethral resection of bladder, ureteroscopic, or hysteroscopic procedures.1,3
- TURP syndrome complicates 10-15% of TURP procedures. Risk factors for TURP syndrome include increased intravesical pressure, prolonged resection, hypotonic irrigation fluid, and open prostatic sinuses.1,3
- TURP syndrome can occur as early as 15 minutes into the procedure and has been reported up to 24 hours postoperatively.3
- Early hypervolemia-related hypertension is often followed by severe hypotension secondary to increased capillary filtration, hypovolemia, decreased cardiac function, and sympathetic blockade. Glycine, if used as the irrigation fluid, may contribute to the visual disturbances and seizures (Figure 2).
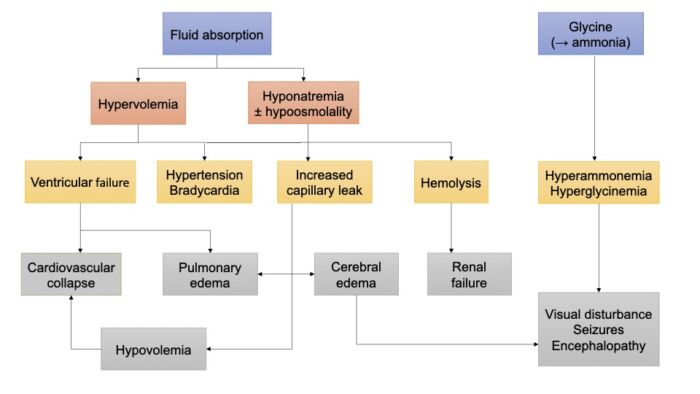
Figure 2. Pathophysiology of TURP syndrome. Adapted from Edwards M, Grocott M. Perioperative fluid and electrolyte therapy. In: Miller RD, editor. Miller's Anesthesia. 9th edition. Philadelphia, PA; Elsevier/Saunders; 2019: 1774-5, 1787-90.
- The signs and symptoms of TURP syndrome are listed in Table 1. Signs of TURP syndrome can be detected earlier in an awake patient under spinal anesthesia. In an anesthetized patient, tachycardia and hypertension may be the only indicators.

Table 1. Signs and symptoms of TURP syndrome in increasing severity. Used with permission from Gravenstein D. TURP syndrome: A review of the pathophysiology and management. Anesth Analg. 1997; 84(2): 438-46.
- Toxicity may also result from solute absorption. In the past, distilled water was used as the irrigation solution, but it led to hemolysis and hyponatremia/hypo-osmolality. Currently, glycine 1.5% or sorbitol 2.7% and mannitol 0.54% are commonly used. Absorption of large amounts of glycine can result in hyperglycinemia, hyperammonemia (from degradation of glycine), and transient visual disturbances.
- The use of bipolar resectoscopes has allowed the use of electrolyte-containing crystalloids as the irrigation fluid.
Prevention of Hyponatremia
- The principal consequence of hyponatremia is life-threatening encephalopathy. This is due to brain edema, which causes elevated intracranial pressures and brain herniation. In moderate to severe hyponatremia (>130mEq/L), elective cases should be postponed until correction can take place.
- The following steps should be taken in the perioperative setting to prevent hyponatremia.
- Avoiding hypotonic fluid replacement
- Limiting postoperative free water to only baseline requirements and replacing sodium losses with isotonic fluid replacement1
- Stopping IV fluid replacement therapy as soon as oral replacement is tolerated1
- During TURP surgery
- Avoiding the use of sterile water as the irrigation fluid
- Minimizing resection time as much as possible (less than 1 hour)
- Using bipolar resection system (instead of unipolar), which allows the use of normal saline as the irrigant
- Fluid absorption should be monitored by comparing the amount instilled with the amount removed. Surgery should be paused, and the patient should be assessed if 750 mL has been absorbed in females and 1000 mL in males.1
- Limiting intravesical pressure to less than 15-25 mmHg (governed by the height of the irrigation bag)
- Sodium levels should be checked in patients with4
- prolonged TURP surgery;
- history of chronic renal failure; and
- patients taking medications which can cause hyponatremia (thiazide diuretics).
Management of Hyponatremia
- The management of hyponatremia depends on the patient’s osmolality as well as volume status. Patients with normal or high serum osmolality need to have a reduction in the nonsodium solute, typically with dialysis.2
Management of TURP Syndrome
- Early recognition is critical.
- The surgeon should be advised to stop the irrigation of fluid, discontinue prostatic resection, achieve hemostasis, and terminate the procedure as soon as possible. If necessary, bipolar resection may be used for the remainder of the procedure.4
- Adequate oxygenation and ventilation must be ensured, and supplemental oxygen should be administered.
- The airway may need to be secured. Early intubation should be considered as edema and laryngeal swelling can be a significant concern that can cause a rapid deterioration of the patient. The lungs should be auscultated for signs of pulmonary edema.4
- Stat serum sodium and serum osmolality levels should be checked. Pseudohyponatremia should be suspected in the setting of normal or elevated osmolality with hyponatremia.4
- Most patients can be managed with fluid restriction and a loop diuretic. Intravenous furosemide 5-20mg bolus may be administered.4
- The hyponatremia should be treated if the patient is symptomatic or the serum sodium is less than 120 mEq/L.
- Hypertonic saline should only be used in the setting of severe hyponatremia with hypo-osmolality due to its high risk of neurologic injury and hypervolemia.
Treatment of Symptomatic Hyponatremia
- In patients with moderate symptoms, hypertonic 3% saline may be started at a rate of 1mL/kg/hr for 3-4 hours, after which sodium levels should be rechecked. The infusion rate should be modified such that the sodium does not increase more than 10mEq/L within the first 24 hours of treatment.
- In patients with severe symptomatic hyponatremia (coma, seizures, sodium less than 120mEq/L), a bolus of 100 mL (or 1.5-2 mL/kg) of 3% saline can be administered over 10 minutes and repeated, if needed. Electrolyte levels should be rechecked every few hours.1
- Rapid administration of hypertonic saline has been associated with an increased risk of central pontine myelinolysis, an osmotic demyelination syndrome.
Treatment of Hyponatremic Patients with Hypoosmolality
- Hypervolemic patients need to have water restriction as well as an improvement in renal and cardiac function, usually in the form of inotropic support and diuresis.2
- Hypovolemic patients require a restoration of their total blood volume, usually in the form of normal saline administration. Additionally, sodium losses must be limited.2
- Euvolemic patients may have SIADH, in which case free water restriction and elimination of precipitating causes should be considered. Vasopressin receptor blocking agents such as conivaptan may also be administered, which have been proven to be safe and effective in the treatment of SIADH.
References
- Edwards M, Grocott M. Perioperative fluid and electrolyte therapy. In: Miller RD, editor. Miller's Anesthesia. 9th edition. Philadelphia, PA; Elsevier/Saunders; 2019: 1774-5, 1787-90.
- Prough DS, Funston JS, Saad AF, et al. Fluids, electrolytes, and acid base physiology. In: Barash PG, editor. Clinical Anesthesia, 8th Edition. Philadelphia, PA; Wolters Kluwer Health; 2017: 1032-42.
- Gravenstein D. TURP syndrome: A review of the pathophysiology and management. Anesth Analg. 1997; 84(2): 438-46. PubMed
- Botz GH. Hyponatremia and hypo-osmolality. In: Gaba, DM, Burden AR, Howard SK, Fish KJ. Crisis Management in Anesthesiology. 2nd Edition. Philadelphia, PA. Elsevier/Saunders; 2015: 221-4.
Copyright Information

This work is licensed under a Creative Commons Attribution-NonCommercial-NoDerivatives 4.0 International License.